Melamine As An Efficient Adsorbent For Mercury Ion Removal
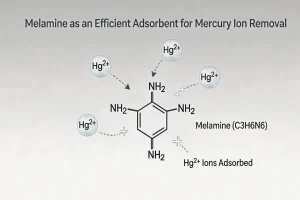
Tech Blog Melamine as an Efficient Adsorbent for Mercury Ion Removal Mercury (Hg) and its compounds are among the most persistent and toxic environmental pollutants.

Urea is one of the most widely used compounds worldwide, with applications ranging from agriculture to automotive emission control. Everyone knows that urea has a high nitrogen content, which may lead many people to ask, ‘Is urea 100% nitrogen?’
Short answer: Urea is not 100% nitrogen. In fact, pure urea contains about 46% nitrogen, making it one of the most concentrated solid nitrogen fertilizers available, but it is not a pure nitrogen compound. To understand why urea is not 100% nitrogen, one needs to look at its chemical structure, composition, and real-world applications – all of which are important for anyone using urea for agricultural, horticultural, or industrial purposes.
Urea is an organic compound with the molecular formula CH, and its molecular formula is CO (NH₂)₂. Its molecular weight is approximately 60.06 g/mol. Urea is one of the simplest organic compounds and the main nitrogen-containing end product of protein metabolism in mammals and certain fish. It was first discovered in human urine in 1773, hence its name.
Pure urea is a white, odorless crystalline powder that is highly soluble in water, ethanol, and benzene. Its melting point is between 131°C and 135°C, with a boiling point of approximately 332.48°C at standard pressure and a water solubility of 1080 g/L at 20°C.
Urea contains four different chemical elements. In a urea molecule, there are:
1 carbon (C) atom
4 hydrogen (H) atoms
2 nitrogen atoms
1 oxygen (O) atom
This elemental composition immediately indicates that urea cannot be 100% nitrogen; it also contains carbon, hydrogen, and oxygen.
To determine the exact nitrogen content of pure urea, chemists use standard percentage composition calculations. This method requires two key pieces of information:
The molecular weight of the entire urea molecule
The total atomic weight of all nitrogen atoms
The formula used is:
%Nitrogen=(total weight of nitrogen atoms ÷ molecular weight of urea) × 100
Let’s calculate using atomic weight in grams per mole (g/mol):
Nitrogen (N): 14 grams/mole
Carbon (C): 12 grams/mole
Hydrogen (H): 1 gram/mole
Oxygen (O): 16 grams/mole
Firstly, calculate the molecular weight of urea (CH ₄ N ₂ O):
Molecular weight=2 (N)+C+4 (H)+O=2 (14)+12+4 (1)+16=28+12+4+16=60 g/mol
Secondly, calculate the total weight of nitrogen atoms in a urea molecule. Because there are two nitrogen atoms:
Nitrogen weight=14+14=28 grams/mole
Finally, apply the following formula:
%Nitrogen=(28 ÷ 60) × 100=46.67%
Correct answer: Approximately 46-47%
In theory, pure urea contains approximately 46.67% nitrogen by weight. In fact, this number has been cited as 46% or 46.6% in industry standards and widely accepted chemical references.
In other words, for every 100 pounds of pure urea, only about 46-47 pounds are elemental nitrogen. The remaining 53-54 pounds are composed of carbon, hydrogen, and oxygen, none of which is elemental nitrogen. Therefore, urea is not 100% nitrogen; It contains approximately 46% nitrogen.
This calculation has been confirmed by multiple authoritative sources, including chemical databases, textbooks, and international fertilizer standards.
The confusion about whether urea is 100% nitrogen may stem from several factors:
Marketing management. Due to urea being the highest-concentration solid nitrogen fertilizer on the market, its high nitrogen content (46%) often attracts attention. This emphasis on nitrogen may lead people to mistakenly believe that urea itself is pure nitrogen.
Incomplete understanding of chemical composition. People who hear that urea is used as a “nitrogen source” may think it is entirely composed of nitrogen, unaware that it is a compound containing multiple elements.
Misunderstanding of fertilizer analysis. Some fertilizers are labeled as containing “100% urea nitrogen”, which means that all nitrogen in the product is in the form of urea rather than other nitrogen forms such as ammonium or nitrate. This is different from saying that the product is 100% elemental nitrogen.
Although urea is not 100% nitrogen, its 46% nitrogen content makes it an efficient and cost-effective fertilizer. The following are the impacts of its use:
-Agriculture: Urea is a neutral fertilizer suitable for all soil types and crops. Its high nitrogen concentration means that less product is needed to provide the same amount of nitrogen as other fertilizers, thereby reducing transportation and application costs. However, urea must be broken down by soil microorganisms (via urease) into ammonium and nitrate for plants to absorb.
-Industrial and other uses: In addition to agriculture, urea is also used in animal feed (as a non-protein nitrogen source for ruminants), medical products (to treat skin diseases), chemical manufacturing (to produce resins and plastics), and even automobile exhaust treatment (to reduce nitrogen oxide emissions). In all of these applications, its nitrogen content is a key characteristic, but its complete chemical composition makes it functional.
Is urea a nitrogen fertilizer?
Yes. Urea is classified as a nitrogen fertilizer because it contains a high concentration of nitrogen (the pure crystalline form accounts for about 46% of its weight) and is applied to crops as a nitrogen source.
Is there 100% nitrogen fertilizer available?
No. All fertilizers contain elements other than nitrogen, even if they are sold at high nitrogen levels. Urea (with approximately 46% nitrogen) is the solid fertilizer with the highest nitrogen concentration commercially available. Other common nitrogen fertilizers include ammonium nitrate (about 34% nitrogen) and urea ammonium nitrate (UAN) solution (28% -32% nitrogen). No solid, stable, and cost-effective fertilizer is 100% elemental nitrogen.
Is higher nitrogen content equivalent to better fertilizer?
Although the high nitrogen content of urea is an advantage, its effectiveness depends on appropriate application (such as deep burial to prevent ammonia volatilization) and soil conditions (temperature and humidity can affect its decomposition rate).
What does the “46-0-0” on a bag of urea mean?
These numbers represent NPK grades – nitrogen (N), phosphorus pentoxide (P) weight percentage ₂ O ₅, potassium oxide (K ₂ O). A bag of urea labeled 46-0-0 contains 46% nitrogen (by weight) and negligible phosphate or potassium fertilizer.
Urea is not 100% nitrogen. Pure urea contains approximately 46.67% elemental nitrogen (by weight), whereas commercial urea typically contains 46.0% to 46.6% nitrogen, depending on purity and grade. This high nitrogen concentration is the highest among all solid fertilizers, making urea a very effective nitrogen carrier, but it is not and cannot be pure elemental nitrogen.
In addition, specific grades of urea used in different applications (agricultural fertilizers, diesel exhaust treatment fluids, animal feed, or industrial resins) have very different purity requirements. Understanding these differences is crucial for selecting products correctly, avoiding expensive equipment damage, and achieving optimal performance, whether it’s fertilizing crops in the field or ensuring emissions compliance on the road.
If you are looking for a high nitrogen fertilizer, urea is a great choice – but remember: its advantage lies in its 46% nitrogen content, not purity. Always check the exact nitrogen content and application instructions on the product label for optimal results.

Tech Blog Melamine as an Efficient Adsorbent for Mercury Ion Removal Mercury (Hg) and its compounds are among the most persistent and toxic environmental pollutants.

Tech Blog Melamine in Polyurethane Foam Application Polyurethane (PU) foam is one of the most widely used polymer materials in the world. They have excellent

Tech Blog Determination of Iron Content in Melamine In the fiercely competitive melamine market, product quality has become a key factor for global customers. Iron

JINGJIANG MELAMINE POWDER
© JINJIANG MELAMINE