
Characteristic analysis of melamine pyrolysis products
Tech Blog Characteristic analysis of melamine pyrolysis products Starting from the organic compound melamine, it was pyrolyzed under vacuum conditions (1000K). The morphology, composition, and

Starting from the organic compound melamine, it was pyrolyzed under vacuum conditions (1000K). The morphology, composition, and structure of the sample were analyzed using X-ray diffraction (XRD), transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS), chemical element analysis (EA), and Fourier transform infrared spectroscopy (FT-IR). The results showed that under vacuum conditions, the complete decomposition product of melamine was a layered graphite phase C₃N₄.
Take 3g of pure melamine powder for analysis, press the powder into small cylindrical pieces with a grinding tool, and place it in a quartz tube. Vacuum the quartz tube, heat it to 1000K under vacuum for 2 hours to fully pyrolyze the sample, and cool the sample to room temperature.
Then remove the yellow powder from the quartz tube wall. The temperature of the area was measured at 500K. Phase analysis of the sample was performed using an X-ray diffractometer, and its morphology and electron diffraction were analyzed using a transmission electron microscope. The binding energy of the sample was determined by X-ray photoelectron spectroscopy, and its chemical bonding structure was confirmed by Fourier transform infrared spectroscopy.
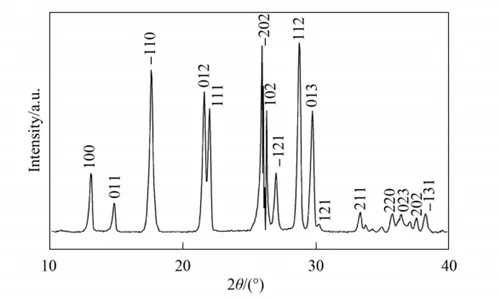
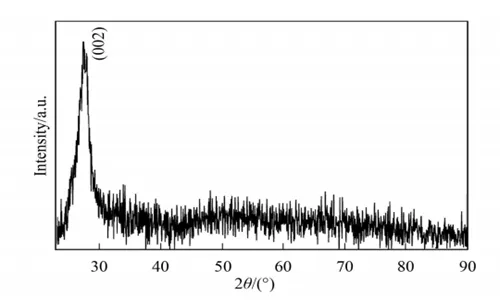
The X-ray diffraction patterns of the raw material melamine and the sample are shown in Figure 1 and Figure 2, respectively. As shown in Figure 2, melamine is completely decomposed, and the decomposition product has a typical characteristic peak at 27.5 °.
This result is consistent with the experimental results, indicating that the obtained sample may be the (002) crystal plane corresponding to the characteristic peak of graphite phase C₃N₄, where d=0.322nm.
The morphology and electron diffraction of the sample were analyzed using Hitachi’s H-8100 transmission electron microscope in this experiment, and the results are shown in Figure 3. As shown in Figure 3, most samples have a layered structure, with some showing a tendency to curl and others already curled into nanotubes at the edges.
From the electron diffraction results, the sample shows a clear diffraction ring, and the d value can be calculated from the ring’s radius. The d value is consistent with the d value of the XRD characteristic peak, indicating that the sample synthesized in this study is graphite phase C3N4 flakes. The elemental composition and chemical bonding states of the sample were analyzed using a Thermo ECSALAB 250 energy spectrometer from the United States. The XPS spectrum of the obtained sample is shown in Figure 4.

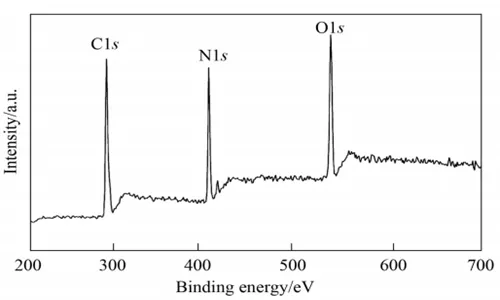
The elemental composition of the sample can be determined through chemical element analysis, and the results are listed in Table 1. As shown in Table 1, the sample mainly consists of carbon and nitrogen, with a small amount of surface-adsorbed oxygen. From the chemical element analysis, the atomic ratio of C to N is 0.72, which is lower than the theoretical value. This may be due to the low nitrogen content caused by the presence of both crystalline and amorphous states in the sample.

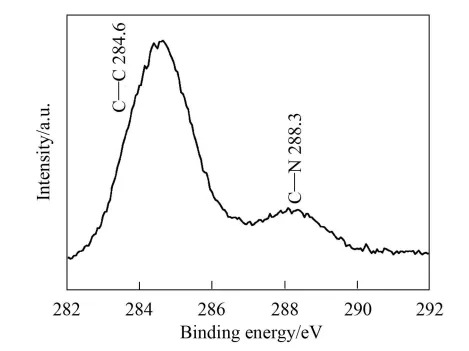
By analyzing the sample’s chemical bonds via XPS, Figure 4 shows that the C1s binding energies are 284.6 eV and 288.3 eV, respectively. The sum of the half-widths of the two peaks is 1.85 eV, which is much larger than the half-width of pure graphite (0.35 eV), indicating that there are at least two binding energies around the C atom. The XPS spectra of samples C1s and N1s are shown in Figures 5 and 6, respectively.
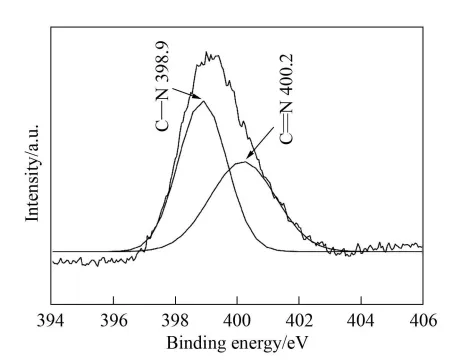
As shown in Figure 5, the peak at 284.6 eV is the standard binding energy of C-C bonds in graphite, and the peak at 288.3 eV is similar to the binding energy of C-N bonds in C1s. As shown in Figure 6, the half-width of N1s is 1.88eV, indicating that there are at least two binding energies around the N atom. Two peaks at 398.9400.2eV were obtained from the fit, corresponding to the binding energies of C-N and C=N, respectively.
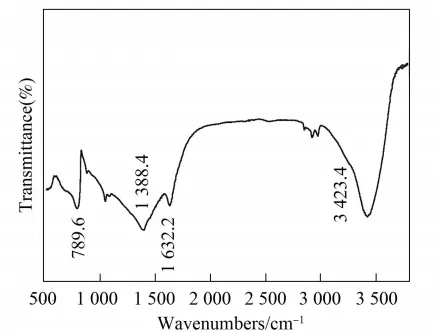
By analyzing the Fourier transform infrared spectrum of the sample, the chemical bond structure can be determined, as shown in Figure 7.
As shown in Figure 7, the absorption peak at 3423.4cm is caused by the NHx (x=1.2) mode, while the absorption peaks at 1388.41632.2cm are caused by the C=N double bond and the C-N single bond, respectively. The absorption peak at 789.6cm corresponds to the amorphous C-C single bond. Consistent with the XPS analysis results.

Tech Blog Characteristic analysis of melamine pyrolysis products Starting from the organic compound melamine, it was pyrolyzed under vacuum conditions (1000K). The morphology, composition, and

Tech Blog Applications of Melamine Flame Retardants in Nylon Melamine flame retardant is a high-performance, halogen-free additive widely used in nylon materials. With excellent flame

Tech Blog Melamine wastewater treatment technology Wastewater generated during melamine powder production contains melamine, ammonium organic acids (amelline, ammelide), ammonia, and other organic pollutants. It

JINGJIANG MELAMINE POWDER
© JINJIANG MELAMINE